ISO 8 Class 100,000 Cleanroom Design and Build
- 2024-02-02
- View 13
ISO 8 or Class 100,000 Cleanrooms and CNC (Controlled Non-Classified) clean spaces common classifications of cleanrooms across a variety of industries and application types. ISO 8 Cleanrooms are utilized in biotechnology, pharmaceutical, electronic assembly, and various cleantech manufacturing applications such as ion lithium battery, solar, surgical kit assembly, packaging, nutraceuticals, and certain packages foods.
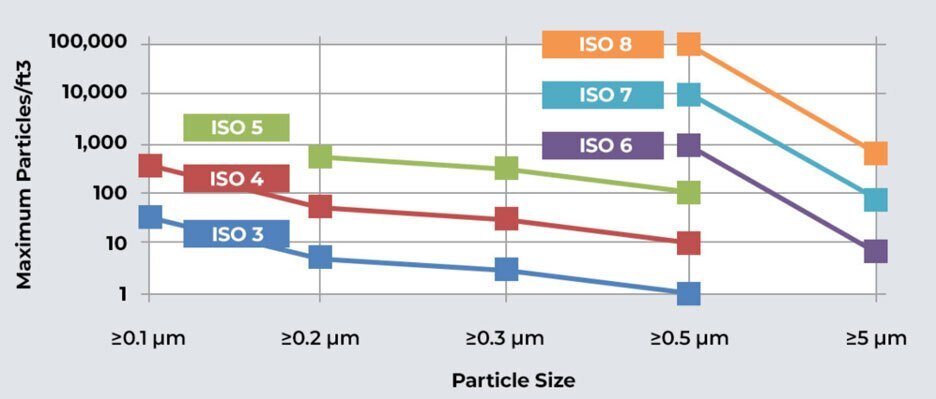
Common ISO 8 Cleanrooms Design Specifications and Considerations
Filtration & Particle Control
Focused measurement of particle sizes greater than 0.5µ and less than 5.0µ
High Efficiency Penetration Air (HEPA) Filtration efficiency: 99.99% at 0.3 microns
Final filtration is performed at the air entry point of the cleanroom and is commonly performed by terminal HEPA modules or HEPA FFU (fan filter modules) depending on the application.
3,520,000 PPC (particles per cubic meter) at 0.5µ
Environmental parameters such as temperature, humidity, lighting levels, noise criteria, static control, and outgassing are often driven by the requirements of the specific process being performed in the cleanroom.
Airflow & Air Change Rates per Hour (ACPH)
Non-unidirectional airflow or mixed air flow patterns typically used. Airflow is most commonly a vertical flow from the ceiling to the floor, returning for conditioning and filtration through low wall returns. Certain applications or zones can make use of an up-flow air flow pattern, although this practice is less common.
Filtration Ceiling coverage is 5-15%, with a randomized array with filtration located over the highest control areas.
Non-unidirectional airflow or mixed air flow pattern typically used.
Architectural Finishes
Wall systems: modular purpose-built wall systems depending on the application of the cleanroom. Biotech applications require non-shedding systems, with monolithic seams and coving to remove 90° angles, allowing wet washed down or VHP fogging to achieve sterile requirements. Nanotechnology and other non-sterile cleanroom types utilize non-shedding systems and are regularly dry wiped or vacuumed.
Ceiling systems: purpose-built ceiling systems are utilized for ISO 8 conditions depending on the application of the cleanroom. Biotech and pharmaceutical applications may use walkable or panelized ceiling systems to promote monolithic washable surfaces, and access to maintain lights and filter media from the top side of the ceiling without breaching the certified and validated space, or gasketed grid systems with lay-in lights and filters. Nanotechnology and other non-sterile applications commonly utilize gasketed grid systems with non-shedding blank ceiling tiles where devices such as filtration or lights are not populated
Flooring systems most commonly a resinous floor system, or heat sealed vinyl flooring system, with specific attributes depending on the application (i.e. chemical resistance, coving, static control, rolling weight loading / impact resistance, non-slip).
Miscellaneous architectural features to include types of doors, viewing windows, glass walls, pass-thrus, sprinkler heads / fire suppression, Wi-Fi / network access, security cameras, and access control devices.
Air returns: low wall returns or ceiling returns.
ISO 8 Cleanrooms Testing Required (ISO 14644-2)
Particle Count Test: maximum time interval 12 months, test procedure ISO 14644-1 Annex A
Air Pressure Cascade: maximum time interval 12 months, test procedure ISO 14644-1 Annex B5
Airflow: maximum time interval 12 months, test procedure ISO 14644-1 Annex B4
Kwang Cleanroom is proud to offer examples of a variety of our cleanroom projects below. Electronics Assembly Cleanrooms, Cosmetic Production Cleanrooms, Automotive Cleanrooms, Aerospace Cleanrooms, Medical Device Cleanrooms, Pharmaceutical & Biotechnology Cleanrooms, Powder Coating Cleanrooms.
