ISO 4 Cleanroom: Maintaining Optimal Cleanliness for Advanced Industries
- 2024-01-13
- View 11
In today's fast-paced and technologically advanced world, industries such as pharmaceuticals, electronics, aerospace, and biotechnology demand environments that are free from contaminants. These industries rely on ISO 4 cleanrooms to ensure their processes meet stringent cleanliness standards. In this article, we will explore the concept of ISO 4 cleanrooms, their importance, and how they contribute to maintaining optimal cleanliness for advanced industries.
1. Introduction
In industries where even the tiniest particle can cause significant damage or contamination, maintaining optimal cleanliness is paramount. This is where ISO 4 cleanrooms come into play. ISO 4 cleanrooms are controlled environments specifically designed to minimize airborne particles, microorganisms, and other contaminants, ensuring the highest level of cleanliness for sensitive manufacturing processes.
2. Understanding Cleanrooms
Cleanrooms are enclosed spaces designed to control and maintain specific levels of airborne particles, temperature, humidity, and other environmental factors. These controlled environments provide a highly regulated space where sensitive operations can be carried out without the risk of contamination.
3. ISO Standards and Cleanroom Classification
To standardize the classification of cleanrooms worldwide, the International Organization for Standardization (ISO) has established a set of guidelines. These standards categorize cleanrooms based on the number of particles allowed per cubic meter of air. The lower the ISO classification number, the cleaner the environment.
3.1 ISO 4 Cleanroom: Definition and Requirements
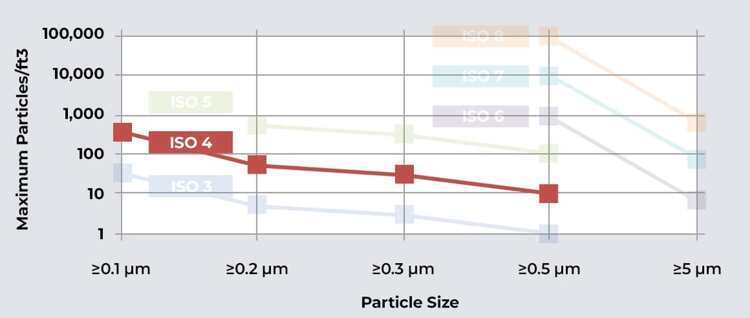
ISO 4 cleanrooms, also known as Class 10 cleanrooms, have stringent requirements for cleanliness. They allow a maximum of 10 particles (0.5 microns or larger) per cubic foot of air. Achieving and maintaining such a high level of cleanliness requires careful design, construction, and operational protocols.
4. Design and Construction of ISO 4 Cleanrooms
Creating an ISO 4 cleanroom involves several crucial elements that contribute to its effectiveness in maintaining cleanliness.
4.1 Environmental Control Systems
ISO 4 cleanrooms rely on sophisticated environmental control systems to regulate temperature, humidity, air pressure differentials, and airflow patterns. These systems ensure that the cleanroom maintains stable and controlled conditions at all times.
4.2 Air Filtration and Purification
The heart of an ISO 4 cleanroom lies in its air filtration and purification systems. High-efficiency particulate air (HEPA) filters are used to remove particles and microorganisms from the air, ensuring a pristine environment.
4.3 Materials and Finishes
The selection of appropriate materials and finishes is crucial to prevent the generation and accumulation of contaminants within the cleanroom. Non-shedding, non-porous, and easy-to-clean materials are preferred to minimize the risk of contamination.
5. Maintaining ISO 4 Cleanrooms
To ensure the ongoing cleanliness and efficiency of an ISO 4 cleanroom, meticulous maintenance and contamination control measures are essential.
5.1 Contamination Control Measures
Strict protocols are implemented to minimize the introduction of contaminants into the cleanroom. These measures include gowning procedures, footwear sanitization, and controlled entry and exit systems to limit the movement of particles and microorganisms.
5.2 Cleaning and Sterilization Protocols
Regular cleaning and sterilization procedures are vital to remove any potential contaminants that may have entered the cleanroom. Specialized cleaning agents and disinfectants are used, following strict protocols to maintain the integrity of the controlled environment.
6. Applications of ISO 4 Cleanrooms
ISO 4 cleanrooms find applications in various advanced industries where precision and cleanliness are paramount. Some notable applications include:
6.1 Pharmaceuticals and Biotechnology
ISO 4 cleanrooms are extensively utilized in pharmaceutical and biotechnology manufacturing processes. These controlled environments provide the necessary conditions for producing drugs, vaccines, and other medical products that must meet strict quality standards.
6.2 Electronics and Semiconductor Manufacturing
The electronics industry heavily relies on ISO 4 cleanrooms for the production of microchips, integrated circuits, and other electronic components. Even the smallest particle can cause significant defects in electronic devices, making the cleanliness of the manufacturing environment critical.
6.3 Aerospace and Defense
In the aerospace and defense sectors, ISO 4 cleanrooms are utilized for various applications, including satellite manufacturing, precision optics, and spacecraft assembly. These industries demand ultra-clean environments to ensure the reliability and performance of their advanced technologies.
7. Benefits of ISO 4 Cleanrooms
The adoption of ISO 4 cleanrooms offers numerous benefits to industries:
Ensures product quality and reliability by minimizing the risk of contamination during manufacturing processes.
Increases yield and reduces defects, leading to cost savings and improved customer satisfaction.
Provides a controlled environment for research and development activities, facilitating innovation and advancements.
Supports compliance with regulatory standards and industry guidelines.
8. Challenges and Considerations
While ISO 4 cleanrooms offer significant advantages, there are certain challenges and considerations to keep in mind:
Cost: Building and maintaining ISO 4 cleanrooms can involve substantial investments due to the need for specialized equipment, materials, and ongoing maintenance.
Operational Complexity: Cleanroom operations require strict adherence to protocols, extensive training, and continuous monitoring to ensure cleanliness standards are met.
Energy Consumption: Maintaining the controlled conditions of a cleanroom can be energy-intensive, leading to higher operational costs and environmental impact.
9. Conclusion
ISO 4 cleanrooms play a vital role in maintaining optimal cleanliness for advanced industries. These controlled environments ensure that sensitive manufacturing processes can be carried out without the risk of contamination. By adhering to stringent cleanliness standards and implementing effective contamination control measures, industries can achieve consistent quality, reliability, and efficiency in their operations.
Kwang Cleanroom is proud to offer examples of a variety of our cleanroom projects below. Hospital Operating Theater Room, Cleanroom Level, Dust-Free Workshop Company, Cleanroom Technician, Buckle Demising Cleanroom, Clean Room Manufacturer, ISO 8 Clean Room.
